Key Concepts
Lee, Zong W. "Unit 2: Atomic & Molecular Structure." YouTube. YouTube, 31 May 2014. Web. 31 May 2014.
Atomic Number
Atoms are made of protons, neutrons, and electrons.
Elements are different because they have different numbers of protons.
Electrons have the same number as protons and the atomic number.
Examples:
Hydrogen = 1 p+ 1 e-
Calcium = 20 p+ 20 e-
Barium = 56 p+ 56 e-
Atoms are made of protons, neutrons, and electrons.
Elements are different because they have different numbers of protons.
Electrons have the same number as protons and the atomic number.
Examples:
Hydrogen = 1 p+ 1 e-
Calcium = 20 p+ 20 e-
Barium = 56 p+ 56 e-
Mass Number
One way to find neutrons is to get the mass number and subtract it by the atomic number or proton.
To find the others do it vice versa:
Mass = proton + neutron
Proton = mass - neutron
Neutron = mass - proton
One way to find neutrons is to get the mass number and subtract it by the atomic number or proton.
To find the others do it vice versa:
Mass = proton + neutron
Proton = mass - neutron
Neutron = mass - proton

<---Element Name
<---Atomic Number/Proton/Electron
<---Element Symbol
<---Atomic Mass
Photo Credit: <a href="http://www.flickr.com/photos/53130103@N05/8672084191/">pennstatenews</a> via <a href="http://compfight.com">Compfight</a> <a href="https://creativecommons.org/licenses/by-nc-nd/2.0/">cc</a>
Isotope
Dalton was proved false about all atoms of the same element being the same.
Although an element may have a different mass number, they are called isotopes.
The mass number could also be after the name of an element:
Nitrogen-15
Nitrogen-14
Iron-55
Dalton was proved false about all atoms of the same element being the same.
Although an element may have a different mass number, they are called isotopes.
The mass number could also be after the name of an element:
Nitrogen-15
Nitrogen-14
Iron-55
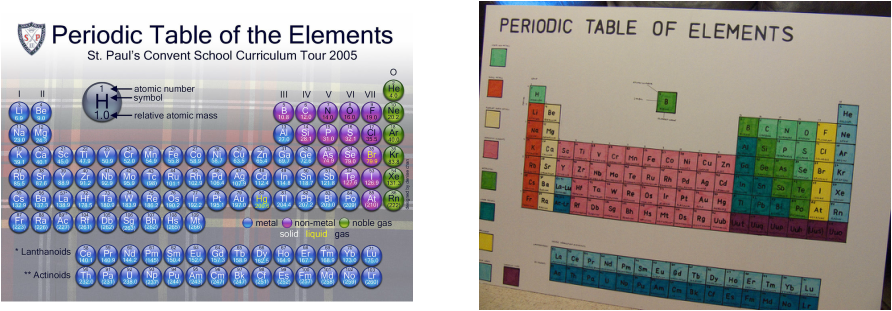
Periodic Table
Mendeleev’s Periodic table is organized in columns in orders of increasing mass similar properties side by side
Moseley arranged element from increasing atomic number (protons)
There are Seven Horizontal Rows or periods.
They're organized by increasing atomic number and identified by a number-letter combination.
The periodic table is organized by chemical properties and groups.
Mendeleev’s Periodic table is organized in columns in orders of increasing mass similar properties side by side
Moseley arranged element from increasing atomic number (protons)
There are Seven Horizontal Rows or periods.
They're organized by increasing atomic number and identified by a number-letter combination.
The periodic table is organized by chemical properties and groups.
Photo Credit: <a href="http://www.flickr.com/photos/42902413@N00/23178762/">denn</a> via <a href="http://compfight.com">Compfight</a> <a href="https://creativecommons.org/licenses/by-sa/2.0/">cc</a>
Photo Credit: <a href="http://www.flickr.com/photos/30070299@N06/4231351150/">poppet with a camera</a> via <a href="http://compfight.com">Compfight</a> <a href="https://creativecommons.org/licenses/by-nc/2.0/">cc</a>
Simple & Weight Averages
For simple averages all you do is find the mean:
4+6+8+10+12=40 Add all the numbers
40/5= 8 Divide it by how many numbers you started with.
The simple average is 8.
For weight averages you multiply the mass with the abundance then add them up:
Carbon-11 with a mass of 11.290 amu and 43.97% abundance
Carbon-12 with a mass of 12.930 amu and 56.03% abundance
11.290 x 0.4397 = 4.96 amu Take the abundance and divide it by 100. Take the mass and multiply it with the abundance.
12.930 x 0.5603 = +7.24 amu Do the same with the other. Now add the results to get the average atomic mass.
12.2 amu
The average atomic mass of Carbon is: 12.2 amu
For simple averages all you do is find the mean:
4+6+8+10+12=40 Add all the numbers
40/5= 8 Divide it by how many numbers you started with.
The simple average is 8.
For weight averages you multiply the mass with the abundance then add them up:
Carbon-11 with a mass of 11.290 amu and 43.97% abundance
Carbon-12 with a mass of 12.930 amu and 56.03% abundance
11.290 x 0.4397 = 4.96 amu Take the abundance and divide it by 100. Take the mass and multiply it with the abundance.
12.930 x 0.5603 = +7.24 amu Do the same with the other. Now add the results to get the average atomic mass.
12.2 amu
The average atomic mass of Carbon is: 12.2 amu
Nuclear Chemistry
In 1896, Antoine Henri Becquerel, Marie and Pierre Curie discovered that uranium emits invisible rays.
Marie Curie called the process radioactivity.
Radiation is penetrates rays that are given off by radioactive sources.
They all won the Nobel Prize in physics for their discovery.
Their theory proved Dalton's idea of invisible atoms wrong.
Isotopes are called radioisotopes and are radioactive, because they have an unstable nuclei.
The nucleus is determined by the protons and neutrons for stability.
If it has too many neutrons, it will lead to an unstable nucleus.
An unstable nucleus loses energy by emitting radiation during radioactive decay.
When an unstable isotope are changed into stable nonradioactive isotopes of a different element it is called transmutation.
There are three main types of radiation which are:
Alpha Radiation Beta Radiation Gamma Radiation
In 1896, Antoine Henri Becquerel, Marie and Pierre Curie discovered that uranium emits invisible rays.
Marie Curie called the process radioactivity.
Radiation is penetrates rays that are given off by radioactive sources.
They all won the Nobel Prize in physics for their discovery.
Their theory proved Dalton's idea of invisible atoms wrong.
Isotopes are called radioisotopes and are radioactive, because they have an unstable nuclei.
The nucleus is determined by the protons and neutrons for stability.
If it has too many neutrons, it will lead to an unstable nucleus.
An unstable nucleus loses energy by emitting radiation during radioactive decay.
When an unstable isotope are changed into stable nonradioactive isotopes of a different element it is called transmutation.
There are three main types of radiation which are:
Alpha Radiation Beta Radiation Gamma Radiation
Photo Credit: <a href="http://www.flickr.com/photos/40936370@N00/5571367545/">Abode of Chaos</a> via <a href="http://compfight.com">Compfight</a> <a href="https://creativecommons.org/licenses/by/2.0/">cc</a>
Half-Life
The level of radioactivity of an isotopes half-life are inversely proportional.
Half-life of a radionuclide is constant.
Disintegration speed doesn't change due to temperature or how much radioactive nuclei is present.
Example:
A 50 gram sample of calcium-20 has a half-life of 20 days. How much calcium-20 will remain in 80 days?
Time| 0 d| 20 d| 40 d | 60 d| 80 d | add up the number of days by itself, until you get to the final day
Mass|50 g|25 g|12.5 g|6.25 g|3.125 g| divide the grams by two until you reach the final day
The amount of calcium-20 that will remain is 3.125 g.
The level of radioactivity of an isotopes half-life are inversely proportional.
Half-life of a radionuclide is constant.
Disintegration speed doesn't change due to temperature or how much radioactive nuclei is present.
Example:
A 50 gram sample of calcium-20 has a half-life of 20 days. How much calcium-20 will remain in 80 days?
Time| 0 d| 20 d| 40 d | 60 d| 80 d | add up the number of days by itself, until you get to the final day
Mass|50 g|25 g|12.5 g|6.25 g|3.125 g| divide the grams by two until you reach the final day
The amount of calcium-20 that will remain is 3.125 g.
Main Source of Information
Cool, Stacey. "Unit 2 Atomic and Molecular Structure." Cool's Chemistry. Merced: S. Cool, 2013. 1-16. Print.